Abstract
Tongue angiosarcomas (TAS) are rarely published mesenchymal tumors of the oral cavity. Despite suitable treatment options such as surgery, chemotherapy and radiotherapy, they are aggressive lethal tumors. There is no globally accepted treatment algorithm for TAS due to scarcity of case reports. In this case report; we presented the first case of giant TAS presenting with both spontaneous bleeding and respiratory distress needing tracheotomy for upper airway obstruction and external carotid artery ligation for bleeding control. With this case report, a detailed review of previous TAS cases was also presented. The risk of abundant bleeding during surgical intervention and need for appropriate arrangements for external carotid artery ligation was emphasized for the surgeons.
Introduction
Sarcomas of the head and neck region are rare malignant tumors. Angiosarcoma (AS) is a cancer of the inner lining of blood vessels. It can be seen anywhere in the body and it is an aggressive malignant tumor with poor prognosis. The most common head and neck regions that AS can originate from are scalp and facial skin, the mandible, maxilla, parotid glands, lips and cheek [1]. The first case report of primary tongue angiosarcoma(TAS) was presented by Dr. Harter in 1927 [2]. Since then, only 8 cases with detailed clinical features were reported in English literature [1-8]. Olson et al. (9) and Fanburg-Smith et al. [10] also presented additional 10 cases, but detailed clinical features( i.e tumor size, initial examination features, detailed treatment protocol and long term follow-up results) were not given. In this case report; we presented the first case of giant TAS presenting with both spontaneous bleeding and respiratory distress.
Case Report
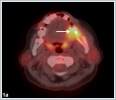
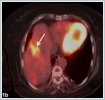
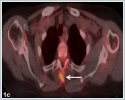
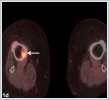
A 65- year- old woman came to the emergency room complaining of intraoral bleeding, and difficulties in respiration. In her past medical history, she had a nodular lesion on her left side of the tongue 3 months ago. The lesion had gradually enlarged. One month ago, she had undergone a Positron Emission Tomography (PET-CT, Siemens Biograph mCT 20, Germany) imaging without pathological diagnosis in another institution. The PET-CT demonstrated a primary malignant tongue cancer with a SUV max of 11.04 and multiple cervical and systemic metastasis involving liver, vertebrae and tibia.(figure 1a,1b,1c,1d)
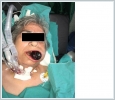
The tumor had abrupt increment in size with spontaneous bleeding. When the patient was evaluated in the emergency room, she had a 6 cm darkish mass on left side of the tongue fulfilling the mouth, also invading the floor of the mouth and tongue base, causing respiratory distress and spontaneous bleeding. (figure 2)
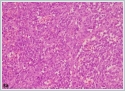
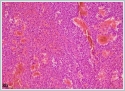
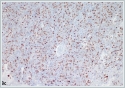
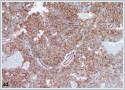
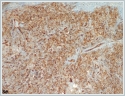
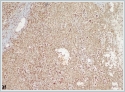
She was immediately taken to the operation room. Tracheotomy was performed under local anesthesia. After tracheotomy, the mass was tried to be excised for pathological investigation under general anesthesia. During surgical invention, abrupt massive bleeding arised from the tumor. Since bleeding could not be controlled with tamponade and bipolar cauterization, external carotid artery ligation was performed and the bleeding diminished. Postoperative histopathological evaluation of the tumor showed thathigh grade spindled tumor cells formed continuous sheets lacking vascular formation in poorly differentiated areas.Tumor cells stained positively for the cluster of differentiation CD31 and CD34 antibodies with immunohistochemical staining, tumor cells also showed positive nuclear staining with Friend leukemia virus integration 1 (Fli1) antibody immunohistochemically (Figure 3a,3b,3c,3d,3e,3f).
These findings were well consistent with TAS. Chemotherapy regimen was planned for the TAS by the medical Oncology department in the postoperative period, but general health status of the patient got worse due to multiple systemic metastasis including lungs. She died at 28[th]day of diagnosis due to liver insufficiency and diffuse lung metastasis causing respiratory failure.
Discussion
Angiosarcoma is a rare malignant mesenchymal tumor comprising less than 10% of all sarcomas. According to our knowledge, 17 cases with TAS have been reported in English literature.
The most common presenting symptoms of TAS are painless or painful tongue mass, tongue swelling and bleeding, and jaw pain. Tumors are generally located in dorsum or lateral part of the tongue [8]. For our case, the patient had a giant left sided tongue tumor causing bleeding and respiratory distress.
The underlying pathogenesis of TAS development is not actually known. Trauma, lymphedema, angioectasia and carcinogen exposure such as vinyl chloride, arsenic and thorium dioxide are presumed to be the contributing factors [1]. Yasumatsu et al. [4] reported a case of TAS arising 30 years after radiation therapy for lymphangioma of the tongue and they thought that previous radiation exposure or radiotherapy may be a risk factor for TAS development. Xeroderma pigmentosum was also speculated to be a contributing factor for TAS pathogenesis [9].
The diagnosis of TAS is primarily made by immunohistochemical investigation. Tumor cells are positively stained for vascular factors such as factor VIII antigens, CD31, CD34, E-selectin and thrombomodulin [1,9 ]. Our case also showed positive staining with CD31 and CD34. Additionally; the tumor cells demonstrated positive staining with another vascular endothelial marker FLI-1.
There is no globally accepted treatment algorithm for TAS due to scarcity of case reports. Surgery is the suitable treatment option for the tumors without systemic metastases and that are amenable to total excision. On the other hand, during surgical intervention the surgeon can encounter an abundant uncontrolled bleeding due to the highly vascularized nature of the tumor, especially for bigger tumors. Rao et al. [5] presented a case of extensive TAS needing bilateral external carotid artery ligation for controlling the bleeding during incisional biopsy. For our case; we also had to perform left external carotid artery ligation to control the massive tumoral bleeding during surgical intervention. Adjuvant radiotherapy may be recommended after surgery. Since TAS has the capability of systemic metastasis by hematogenous route, routine neck dissection is not recommended [1,2,3,7]. For the tumors having systemic metastasis, chemotherapy regimen including recombinant interleukin-2, carboplatin and pirarubicinare recommended in the treatment protocol [4,6] Despite appropriate treatment options, TAS are aggressive lethal tumors with poor prognosis. In case of our case, we performed the surgery for upper airway obstruction and tumoral bleeding. At initial admission the patient had already systemic metastasis. We could not apply any chemotherapy regimen due to additional lung metastasis to previous metastasis during follow-up and bad health conditions not permitting the onset of the chemotherapy protocol.
References
- Chadha V, Rawat DS, Prasad B. Primary angiosarcoma of the tongue: a case report. Int J Otolaryngol Head Neck Surg 2012; 1:51–52.
- Harter JH. Angiosarcoma of tongue: case report. Laryngoscope 1927;37:869–871.
- Frick WG,, McDaniel RK. Angiosarcoma of the tongue: report of a case. J Oral Maxillofac Surg 1988;46:496–498.
- Yasumatsu R, Hirakawa N, Tomita K. Postradiation angiosarcoma of the tongue. Eur Arch Otorhinolaryngol 2000;257:464–465.
- Rao KK, Kumar HN, Rao RV. Primary haemangiosarcoma of the tongue. Int J OralMaxillofac Surg 1986;15:489–491.
- Tabata M, Sugihara K, Matsui R, Yonezawa S, Abeyama K, Maruyama I.Angiosarcomaof the tongue: report of a case with immunohistochemical findings. J Oral Pathol Med.1999;28:92-95.
- Nagata M, Yoshitake Y, Nakayama H, Yoshida R, Kawahara K, Nakagawa Y. Angiosarcoma of the oral cavity: a clinicopathological study and a review of the literature. Int J Oral Maxillofac Surg. 2014;43:917-23
- Patel PB, Kuan EC, Peng KA, Yoo F, Nelson SD, Abemayor E. Angiosarcoma of the tongue: A case series and literature review. Am J Otolaryngol. 2017;38:475-478
- Olson MT, Puttgen KB, Westra WH. Angiosarcoma arising from the tongue of an 11-year-old girl with xeroderma pigmentosum. Head Neck Pathol 2012;6:255–257.
- Fanburg-Smith JC., Furlong MA., Childers ELB., Oral and Salivary Gland Angiosarcoma:A Clinicopathologic Study of 29 Cases. Mod Pathol.2003 ;16:263-271.
|