|
|||||||||
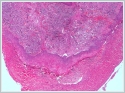
Turkish abstractNöroendokrin karsinomlar (NEC'ler), iyi diferansiye tümörlerden yüksek dereceli tümörlere kadar değişen çok çeşitli bir malign epitelyal nöroendokrin neoplazm grubu oluşturur. Nöroendokrin tümörlerin sınıflandırılması; gastrointestinal sistem tümörleri, pankreas, baş ve boyun, deri ve akciğerler tümörleri olarak lokalizasyonlarına göre tarif edilmiştir. Diğer önemli olan, birçok klinisyen ekstrapulmoner NEC'leri sınıflandırmak için Dünya Sağlık Örgütü (WHO) pulmoner kılavuzlarını kullanır. Dünya Sağlık Örgütü şu anda, mitotik oran ve nekrozun yaygınlığına dayalı olarak dört ana akciğer nöroendokrin tümör tipini tanımaktadır: tipik ve atipik karsinoidler ve küçük ve büyük hücreli NEC'ler. Alt tip, prognostik ve terapötik öneme sahiptir.IntroductionNeuroendocrine carcinomas (NECs) in the head-and-neck area, including the larynx, are rare, but well documented [1-4]. Most patients with NECs are heavy tobacco users, and more than 90% of NECs occur in the supraglottic region [1,5,6]. An association with human papillomavirus has been identified, but may not be as significant as the associations with oropharyngeal and sinonasal tract tumors [1,7,5]. Patients with NECs present with non-specific symptoms, including hoarseness and dysphagia [8, 9]. In the World Health Organization’s (WHO’s) 2017 Blue Book, NECs are classified as well differentiated, moderately differentiated, and poorly differentiated, and two subtypes (small-cell and large-cell neuroendocrine carcinomas [SmCNECs and LCNECs]) and paragangliomas (NECs of neural origin) are recognized [1]. However, some authors consider laryngeal LCNEC to be a separate entity, given its aggressive behavior. Case ReportA 59-year-old male presented with hoarseness. His past medical history was unremarkable. Laryngoscopy was used to examine the vocal cords. The left arytenoid protruded into the lumen and was leukoplakic over an area of 1 × 1 cm. A tumor presented as a submucosal fleshy polypoid, which had been partially distorted by the forceps. On thoracic computed tomography, a spicular lesion with a length of approximately 9 mm and a pleural recess was evident in the left lung apex, and was compatible with scar tissue. Follow-up was recommended to exclude malignancy. After biopsy from the larynx a few burgundy-colored tissue samples in a single cassette; the largest was 1.5 × 0.6 × 0.6 cm and the smallest was 0.1 × 0.1 × 0.2 cm. Histomorphologically, the tumor exhibited prominent cell nests with organoid patterns in certain areas (Figure 1).
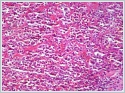
The cells exhibited slight prominent nuclear pleomorphism, a moderate amount of eosinophilic cytoplasm, vesicular chromatin, clear nuclei, and prominent nucleoli (Figure 2). The maximum mitotic count was 2/10 high-power fields; focal necrosis was also noted.
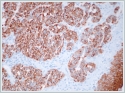
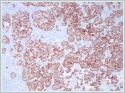
Immunohistochemistry confirmed that the tumor was neuroendocrine in nature. Tissue samples were diffusely positive on staining for pancytokeratin (Figure 3), chromogranin A, synaptophysin (Figure 4), and CD56. The samples did not stain for calponin, anti-S-100 protein, or TTF-1; the extent of Ki-67 staining was greater than 20%.
Negative S-100 status helpfully distinguishes moderately differentiated NECs from malignant melanomas and paragangliomas. The nest pattern of a moderately differentiated NEC may be confused with clusters of melanoma cells; NECs are known to mimic several other tumors occurring in the head-and-neck region [10]. A thorough clinical and radiographic work-up revealed no other primary or metastatic disease, confirming the diagnosis of a primary laryngeal NEC. DiscussionTumors of the neuroendocrine system are heterogeneous, varying in origin, nomenclature, classification, location, histological appearance, extent of differentiation, biological behavior, and functional activity, but share certain immunohistochemical and ultrastructural characteristics.Previous reports have highlighted the predominance of moderately differentiated NECs (atypical carcinoids) in the larynx, whichconstitute 0.2–0.6% of all laryngeal malignancies. Goldman et al. (1969) first described atypical laryngeal carcinoids. These tumors occur in heavy smokers in their 60s, and the male:female ratio is 3:1 [11]; most (90%) tumors occur in the supraglottis, usually in the aryepiglottic fold, arytenoid, and epiglottis [12]. The presenting symptoms vary by tumor location and include hoarseness, dysphagia, pharyngodynia, hemoptysis, and dyspnea. The pathological features include nests of monomorphic cells with round nuclei and speckled chromatin; occasionally, moderate atypia is evident. NECs are immunohistochemically positive for chromogranin, synaptophysin, CD56, and NCAM. A well-differentiated NEC (a carcinoid) can be treated by local excision; a moderately differentiated NEC (an atypical carcinoid) is best treated surgically, and both types of poorly differentiated laryngeal NEC seem to respond optimally to chemotherapy. Some authors consider that laryngeal LCNEC is a newly defined clinicopathological entity; its treatment has not been standardized. Whether cases of this tumor occurring in the lung should be treated as SmCNECs or different tumors remains controversial [13]. The recommended management is complete surgical removal with partial or total laryngectomy, depending on the tumor size and extension. As the risk of cervical lymph-node involvement is high, bilateral elective neck dissection should be performed. Some authors maintain that limited dissection (at levels IIA and III) is sufficient for patients lacking clinical lymphadenopathy [5]. The 5-year disease-specific survival rates are 100% for patients with well-differentiated NECs (carcinoids), 53% for those with moderately differentiated NECs (atypical carcinoids), 19% for those with SmCNECs, and 15% for those with LCNECs. LCNECs are very aggressive; most patients develop distant metastases and die within 2 years (13). In one study, the 5-year survival rate of patients with laryngeal SmCNECs was less than 10%. In another study, the 5-year survival rate of patients with atypical carcinoid tumors of the larynx was about 46.7%. Our patient’s clinical symptoms were typical of a laryngeal NEC. The primary tumor was located in the arytenoid, as expected. The histological features and immunohistochemical data were characteristic of a moderately differentiated NEC. The pathological findings included synaptophysin, chromogranin, Ki67, pancytokeratin, and CD56 positivity. The maximum mitotic count was 1 to 2/10 high-power fields. Tumor necrosis was patchy and focal; we found no vascular or perineural invasion. Although distant metastasis is common in patients with laryngeal NECs, we found no such metastasis in our patient.
References
|
|||||||||
| Keywords : larinks , aritenoid , nöroendokrin karsinomlar , baş ve boyun | |||||||||
|