|
|||||||||
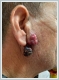
AbstractRenal cell carcinoma is the most common malignant tumor of the kidneys. When the renal cell carcinoma is diagnosed, 30% of patients have metastasis to distant organs such as lungs, bones, liver, lymph nodes and mediastinum. Metastasis to the head and neck region is very rare. In these case reports, first patient is a 64-years-old male who had a right radical nephrectomy operation for renal cell carcinoma 20 years ago and presented with a mass in the right parotid region and second patient is a 63-years-old male who had a left nephrectomy operation 10 years ago due to renal cell carcinoma and presented with a recently developed mass lesion on the left tonsil. In this case report series, we aimed to present the clinical features and treatment strategy of head and neck metastases in renal cell carcinoma, which is a rare condition.IntroductionRenal cell carcinoma (RCC) constitutes 3% of all adult malignant tumors. It is more common in men and in the third to sixth decades of life. The clear cell is the most common histological variant of RCC [1]. Clear cell carcinomas constitute 80-85%of the adult renal malignant tumors. Smoking, hypertension and obesity are the main risk factors [2]. 30 to 40% of the RCC patients have distant metastases at the time of diagnosis [3]. Moreover 10% of the patients present with metastasis before clinical diagnosis of the primary tumor. Local recurrence or metastasis of the primary tumor develops in approximately 50% of the patients during the treatment and follow-up [1]. Lungs, bones, liver, and brain are the most common metastasis regions [3]. Metastasis to the head and neck region is very rare (less than 1%) [4]. Otherwise, 8-15% of all head and neck region metastasis are associated with RCC [1]. 5-year survival has been reported to be between 0% and 20% in patients with head and neck RCC metastases [5]. RCC distant metastasis occurs through the lymphatic, hematogenous or Batson's venous plexus. Batson's venous plexus is a paraspinal venous plexus causing metastasis by tumor embolism to the head and neck region without lung involvement [6]. In these case reports, head and neck metastasis of RCC were presented with the current literature. Case ReportFirst patient is a 64-year-old male who admitted with a progressive swelling in the right parotid had two protruding lesions on the parotid region skin. The lesions were 2 cm in size (Figure 1).
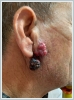
Facial nerve functions were normal and there was no other palpable swelling in the neck. Magnetic resonance imaging (MRI) of the neck revealed 16x10 mm two solid lesions which were located subcutaneously and involving the superficial parotid gland (Figure 2).
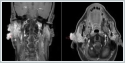
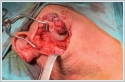
Positron emission tomography-computerized tomography (PET / CT) imaging revealed 33x26 mm hyper metabolic (SUV max: 4.9) mass in the right parotid region with irregular borders. No other pathological involvement was observed in the rest of the body. The patient had right radical nephrectomy due to RCC, 20 years ago and received 6 cycles of cisplatin, carboplatin and etoposide chemotherapy owing to small cell carcinoma originating from the upper lobe of the right lung, 4 years ago. Fine needle aspiration biopsy (FNAB) was performed for the parotid lesions and reported as renal cell carcinoma metastasis. Total parotidectomy including skin lesions covering the parotid gland were performed (Figure 3).
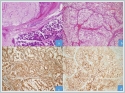
Facial nerve main truncus and its branches were preserved. Skin defect was repaired primarily. The lesion was reported as renal cell carcinoma metastasis with negative surgical margin (Figure 4). Histopathological examinations revealed that the tumor metastasis originated from the subcutaneous tissue in the parotid region and extended to the parotid gland. The patient received localized postoperative adjuvant radiotherapy treatment for the tumor due to the absence of involvement in another part of the body in PET / CT imaging. In addition, sunitinib, a tyrosine kinase inhibitor, was initiated as postoperative adjuvant therapy to prolong overall survival and disease-free survival. No recurrence or metastasis was observed in the imaging of the neck, thorax and abdomen during the follow-up of the patient. Second patient is a 63-year-old male who admitted to the emergency department with a complaining of intermittent blood flow from the mouth and dysphagia for two days was consulted to the head and neck department. Endoscopic larynx examination revealed almost 3 cm pedunculated brownish hemorrhagic mass attached to the lower part of left palatine tonsil. No other lesion was observed in the hypopharynx and larynx and no palpable mass on the neck. Patient was operated urgently due to the high risk of upper respiratory tract obstruction and bleeding. The lesion was totally excised with peduncle from the tonsil with macroscopic clean surgical margins. The patient had left nephrectomy operation and chemotherapy treatment 10 years ago due to clear cell-RCC. The histopathological examination of the tonsillar lesion was reported as clear cell renal carcinoma metastasis. A PET / CT imaging revealed metastatic lesions with increased FDG uptake (SUD Max: 3.9-4.2) in the right supraclavicular region, mediastinum, lung and left testicle. Tonsil tissues were normal without any increased FDG uptake. Chemotherapy was initiated after surgery in 2019 and the treatment continues. PET-CT Scan in 2021 shows numerous hypermetabolic LAPs in the mediastinum, metastatic nodular-masses in both lungs, metastatic pleural lesion that invades the third rib and hypermetabolic lymph node in the abdomen.
DiscussionParotid gland is the most common region which the salivary gland tumors arise [7]. The most common benign parotid tumor type is pleomorphic adenoma (60%), followed by Warthin tumor (10%) [8]. The most common malignant tumor is mucoepidermoid carcinoma and commonly originates from the parotid gland. On the other hand, metastasis to the parotid gland is most commonly caused by malignant melanoma (45%), followed by squamous cell carcinoma in the head and neck region (37%) [9,10]. Although RCC has a potential to metastasize to atypical tissues or organs, there are quite a few case reports in the literature regarding metastasis to the parotid gland [11]. RCC is divided into 4 subtypes, these are clear cell, papillary, chromophobe, and collecting duct tumors. The most of cases (75%) are clear-cell RCC [12]. The 5-year survival rate of RCC patients after nephrectomy is between 60 and 75%. Survival rate decreases in patients with metastases. Metastasis to a single site with radical treatment, survival rate is reduced to 35%. As the number of metastases increases, survival rate decreases to 0–7%. Single site metastases that develop after treatment of primary tumor have a better prognosis than the metastases that present during primary disease. The 5-year survival rate in these patients is approximately 50% [13]. According to the literature, parotid gland metastasis may occur before or after primary therapy for RCC [12] In the case series of Anja Lieder et al., 671 consecutive RCC patients were evaluated retrospectively and 200 (30%) of the patients were found to have metastasis after the diagnosis in twenty-four months follow-up. They detected distant metastases in 172 patients and 22 (3.3%) patients had metastases in the head and neck region. They were mostly detected in cervical and cranial lymph nodes, then in the parotid and thyroid gland, tongue, forehead skin, skull, and paranasal sinuses. All head and neck metastases were treated by surgical excision, 14 patients received adjuvant radiotherapy while 9 patients received chemotherapy or targeted therapy. In 19 of 22 patients, they detected simultaneous disseminated metastases in other organs at some stage of the disease course. In 3 of 22 patients, metastasis occurred only in the head and neck [14]. In both of our cases, metastatic lesions in the head and neck region were surgically excised, one of them had multiple metastasis and chemotherapy was the single treatment modality after surgery but the other patient had the single site metastasis and he received radiotherapy and targeted therapy after surgery and no recurrence was observed during follow-up. RCC is known as a radiation-resistant tumor but radiotherapy reduces tumor volume and plays an important role in controlling symptoms associated with pain and local compression [15]. On the other hand, malign tonsil tumors are known to constitute75% of all tonsil tumors [16]. Primary tonsil malignancies constitute 2% of all malignancies. Squamous cell carcinomas (75%) are the most common primary tumors of tonsils while lymphomas take the second place. Tonsillar tissue malignancies are generally seen as primary tumors. Metastatic involvement of tonsillar tissue is extremely rare (0.8%), and it often originates from the breast, skin, kidney and colorectal system through lymphatic spread [17]. RCC metastasis in tonsils have been reported in a very few cases before [17]. Dario Marcotullio et al. reported a RCC case with tonsillar metastasis who presented with complaints of oral bleeding and dysphagia similar to case that was presented in the study. This patient had undergone nephrectomy for RCC 3 years ago and there was no sign of metastasis. He was given adjuvant radiotherapy after tonsillectomy and no recurrence was observed in the 6-month follow-up. [18]. Bom JW at all determined survival rates reduces significantly and tonsillectomy and additional treatment (radiotherapy, chemotherapy) need in RCC’s tonsil metastasis in their review of literature [19]. Menauer et al suggested radical resection of tonsillar metastasis [20]. In the case we presented, tonsillectomy was not performed. Operation was performed in emergency condition and the first thing to do was stop the bleeding and exicion of the lesion. The surgical margin was negative. There is no metastatic tumor was observed in tonsil in subsequent PET / CT imaging. Because of the patient had systemic involvement and there is no tumor left in tonsil, additional radiotherapy was not applied to the tonsillar area. Postoperative chemotherapy treatment was given because of the widespread metastasis in the body. No pathological involvement was observed in the tonsillar region in PET/ CT imaging during follow-up as well. Considering that the patient has systemic metastases and need systemic treatment, no more surgical treatment was performed. Conclusion There is not a consensus about the treatment of head and neck metastases of RCC. Treatment should be chosen according to the affected area and the general condition of the patient. Surgery is recommended as the primary treatment method, especially in patients without other organ involvement [4]. On the other hand radical surgical procedure may not required in systemic metastases. RCC has traditionally been defined as a radiation resistant tumor. Therefore, the role of radiotherapy as a primary approach is controversial [21]. Patients should evaluate multidisciplinary in terms of postoperative treatments after surgical operations. It should be kept in mind that in patients with a history of RCC, metastatic lesions may be seen in atypical locations in the head and neck region. Informed ConsentFrom the PatientReferences
|
|||||||||
| Keywords : Renal hücreli karsinom , baş boyun , metastaz , parotis bezi , palatin tonsil | |||||||||
|