|
|||
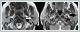
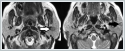
AbstractSalivary gland neoplasms make up 3%-4% of head and neck neoplasms. Salivary gland neoplasms are present most often in the parotid gland. It is a rare case to see synchronous salivary gland tumors with different histological type at the ipsilateral salivary gland. In this study, the diagnosis and treatment process of a patient with an unrelated synchronous warthin tumor and basal cell adenoma combination in the ipsilateral parotid gland was described.IntroductionSalivary gland neoplasms make up 3%-4% of head and neck neoplasms. Almost 80% of salivary gland neoplasms are present in the parotid gland [1]. Warthin tumor (WT) is predominantly seen in parotid gland. WT is the second most common benign salivary gland tumor after pleomorphic adenoma (PA) [2]. It occurs more commonly in males than in females [3]. Diagnosis by fine needle aspiration biopsy is often possible and may be used as a guide for operational treatment [4]. Basal cell adenoma (BCA) is a rare benign epithelial tumor of salivary gland. BCA localises most often in parotid gland. BCA is composed of uniform basaloid cells and named from the basaloid appearance of these cells [5]. Synchronous ipsilateral salivary gland tumors with different histological type are rare in literature [6]. Most of the tumors that develop this way are seen in parotid gland. While WT is described as the most common neoplasm among synchronous parotid gland tumors, WT in conjunction with PA is described as the most frequent synchronous parotid tumor combination [7]. Herein we aimed to present a case of synchronous ipsilateral WT and BCA with relevant radiologic, cytopathological and histopathological findings. Case ReportA 60 years old female tobacco smoker patient presented to our institution with a slowly enlarging, painless mass for three years in left parotid gland. Co-morbidities were; hypertension, coronary artery disease and allergic asthma. Ultrasonography of the left neck demonstrated two cystic spheric lesions of 13 mm and 8 mm in diameter at left parotid gland. In addition, magnetic resonance (MR) of the neck revealed two lesions, measured 8.5x8.8 mm in superficial lobe of the parotid gland and 13.3x10.2 mm in deep lobe of the gland which were well-circumscribed, hypodense in T1A series and slightly hyperdense in T2A series (Figure 1).
The mass in the superficial lobe was denser than the normal paranchyma and the mass in the deep lobe was undistinguishable. Subsequently biopsy of each mass was planned. Ultrasound guided fine needle aspiration biopsy of the each solid component was performed. The superficial mass was diagnosed as salivary gland neoplasm in tubular pattern (though it is not the final diagnosis, BCA and low grade adenoid cystic carcinoma were considered as differential diagnosis) and the mass in the deep lobe was resulted with findings relevant to WT. Based on imaging and cytopathological findings total parotidectomy was performed with dissection and preservation of the facial nerve by using intraoperative facial nerve monitorization. On the third postoperative day, when blood less than 20 cubic centimeters (cc) were detected from the drain, the drain was removed and the patient was discharged. Compressed dressing was applied to the patient for 7 days. No postoperative complications were detected. Two different masses were seen in the superficial and deep lobe of the total parotidectomy material. The superficial mass was 1.1 cm and the deep mass was 1.5 cm in diameter. The mass in the deep lobe, a predominantly solid, well-circumscribed lesion composed of ribbons of double-layered oncocytic cells and a lymphoid stroma with germinal center formation was diagnosed as WT. Other mass a predominantly cystic lesion, composed of basaloid cells and deposits of membrane like materials was diagnosed as BCA. No residual tumor was seen in surgical margin after resection. DiscussionThe incidence of salivary gland neoplasms are 1-2/100.000 [8]. More than 30 salivary gland tumors are listed in the World Health Organization salivary gland classification [9]. Most common benign tumors of salivary glands are PA and WT. Smoking was reported as a substantial risk factor for development of WT [10]. No relation was reported between other salivary gland neoplasms and smoking [11]. The incidence of bilateral WT in the literature has been found to be close to 10 %. [12,13]. Our case supports the information that smoking of the patient is an important risk factor for the development of WT. BCA is one of the rare salivary gland tumors and occurs predominantly in parotid gland. It can occur at every age more commonly in seventh decade. Both of the genders affected equally [5]. BCAs are usually smaller than 3 cm, presented as slowly enlarging, asymptomatic mobile masses [14]. BCAs are histopathologically classified into four subtypes as; solid, trabecular, tubular and membranous [10]. Presence of multiple tumor formation within parotid gland is a rare event. Reports of various studies that evaluate 5,000 patients with parotid gland tumor revealed that this occurs only in 0.4-0.7% of the cases. At that, multifocal WT, PA, BCA and oncocytoma are the most common [7]. Zeebregts and colleagues assessed 341 patients who have undergone parotidectomy for resection of salivary gland lesions. 14 of 341 patients have two or more synchronous tumor in ipsilateral salivary gland. The most common combination was multiple WT. PA/WT combination was in the second order after ipsilateral synchronous salivary gland tumor. No patient was with synchronous ipsilateral WT and BCA parotid tumor [15]. Schilling et al. reported WT were found in association with another tumor in 24 of 33 reported cases (82%). But they did not detect synchronous WT with BCA [16]. Gnepp and colleagues reported 25 cases of synchronous ipsilateral salivary gland tumors. One of the cases was a 77 year old male patient with WT and BCA. Mean age of the cases were 63.9 years. Male and female ratio was 9:1 [7]. Though synchronous parotid tumor was reported more commonly in males our patient was female. In this case, MR imaging of the neck, ultrasonography of the parotid gland, cytopathologic and histopathologic evaluation demonstrated solid and cystic areas which were diagnosed as two separate masses. Together with some radiologic studies, this case also showed the importance of imaging in the detection of potential synchronous parotid gland lesions and rigorous sampling [17]. As a result, development of ipsilateral synchronous tumor in salivary gland is a rare case. When separate lesions are detected in examination and imaging methods, biopsy should be performed from all separate lesions. In such, the presence and the types of synchronous tumors should be determined with pretreatment imaging methods and cytopathological evaluation and the extent of the operation should be decided. References
|
|||
| Keywords : Biyopsi , neoplasm , parotis bezi. | |||
|